Two new breakthrough treatments brings fresh possibilities for those living with chronic plaque psoriasis.... Read more >

Help us make an effective psoriasis treatment more accessible
Help us make an effective psoriasis treatment more accessible
Apremilast (Otezla®) is an oral medication used to treat chronic plaque psoriasis (CPP) and psoriatic arthritis (PsA). Currently, only dermatologists and dermatology registrars can start a patient on apremilast but these specialists are in short supply.
Published 8 September 2023

What is chronic plaque psoriasis?
Psoriasis is known for symptoms you see on the skin, but this condition runs deeper than that. It is an autoimmune disease that occurs when the immune system starts attacking healthy cells — usually skin cells — instead of targeting foreign bodies like bacteria and viruses.
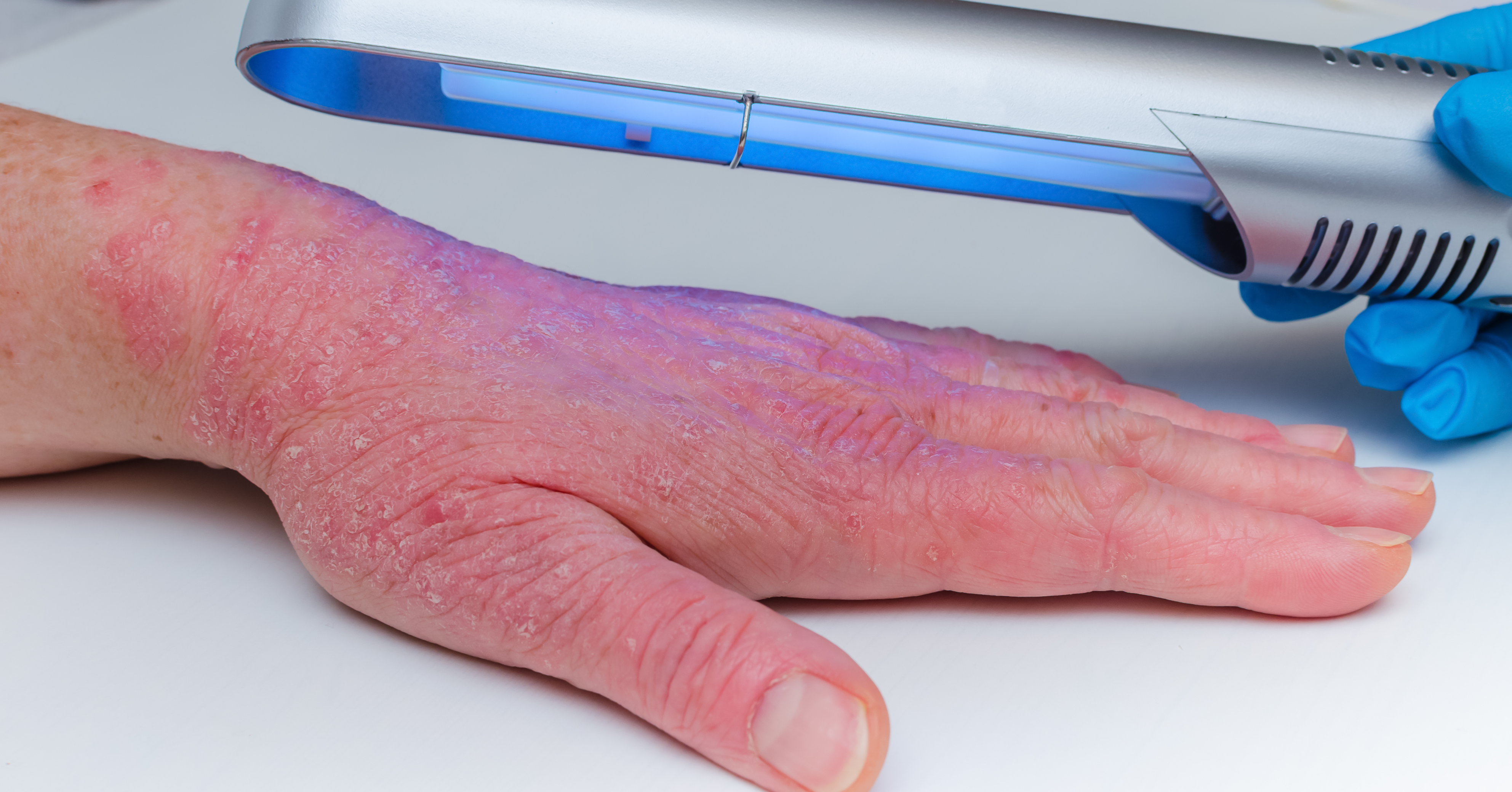
What sets psoriasis apart from typical rashes are the thick white or silvery patches that are the classic tell-tale marker of a type of psoriasis called plaque psoriasis. These plaques (also called lesions) usually show up on the elbows, knees and scalp, but they can appear anywhere on the body. Plaque psoriasis occurs in 90 per cent of people affected by the condition.
Plaques can occur on different parts of the body including the face, scalp, hands and feet, groin, skin folds and nails. They can be very dry, cracked and itchy but scratching can cause them to bleed. They vary from mild patches in a few spots to severe cases that cover most of the body.
There is currently no cure for psoriasis but it can be well-managed with appropriate proven treatment.
What is apremilast?
Apremilast is a type of disease-modifying antirheumatic drug called an oral phosphodiesterase 4 (PDE4) inhibitor. In Australia, it is used to treat chronic plaque psoriasis and psoriatic arthritis.
PDE4 is a natural substance in the body’s cells that helps to regulate immune activity associated with psoriasis and psoriatic arthritis. Managing this immune activity helps to control the symptoms of these conditions.
In clinical trials, apremilast effectively reduced the severity of different dermatological manifestations of psoriasis and improved patients’ quality of life. This means that the impact of the condition on daily activities, relationships and other factors should be less than it was before.
Apremilast has an acceptable safety profile and is generally well-tolerated. It is taken twice a day in tablet form.
Apremilast and the Pharmaceutical Benefits Scheme
In Australia, the apremilast access requirements for people with chronic plaque psoriasis are currently as follows:
- The patient must be at least 18 years of age.
- The patient must have severe CPP that significantly interferes with their quality of life and have failed treatment with, or are contraindicated or intolerant to, methotrexate.
- The patient must not be undergoing concurrent PBS-subsidised treatment for psoriasis with a biological medicine or ciclosporin.
At present, only dermatologists and dermatology registrars (in consultation with a dermatologist) can start a patient on apremilast through the PBS. General practitioners (GPs) can continue treatment under the direction of a dermatologist or dermatology registrar.
However, there aren’t enough dermatologists or dermatology registrars in Australia. There are only 600 dermatologists to meet the needs of nearly 26 million Australians (that is 1 for every 43,000 people).
This can make access to specialty dermatology care difficult, especially for patients in rural and remote areas or those who largely rely on the public healthcare system and general practice. In many cases, patients can wait months or even years for an initial consultation with a dermatologist in Australia.
The Pharmaceutical Benefits Advisory Committee (PBAC) has been asked to consider:
- Adding rheumatologists and general physicians as prescribers; and
- Allowing GPs to start patients on apremilast if they are experienced in the treatment of psoriasis or have consulted an experienced prescriber.
What does this mean for Australians with CPP?
These changes aim to help more patients access treatment, particularly those that have difficulty accessing specialty care. For example, many patients in rural and remote areas have to travel long distances and pay for accommodation in order to see a specialist. Even those in urban areas can struggle to afford specialist appointments. Therefore, accessing apremilast through their GP would be far less time consuming and costly for such patients.
Expanding the list of health professionals who can initially prescribe apremilast could also help patients start treatment much earlier in their psoriasis journey. For many, this would mean less time dealing with painful and unsightly symptoms and the social stigma that often accompanies them.
What we’d like you to do
The PBAC welcomes submissions from patients, carers, health professionals, consumer groups or organisations and members of the public on medicines submitted for PBAC consideration.
GHLF Australia would like to collate your comments into a single submission and send it to the PBAC on your behalf. Your comments can respond to any or all of the following questions:
- What is the impact of psoriasis on your life? Try to be as specific as possible including impacts on your everyday activities, work, family, friends, and mental and emotional health.
- What are some of the barriers you have encountered when trying to access effective treatment for your psoriasis? How have those barriers impacted you?
- What do you see as the advantages of expanding the list of who can initially prescribe apremilast? For example, what impact could it have on your condition or quality of life?
- What do you see as the main disadvantages of this proposed change?
You are welcome to provide any additional comments you would like the PBAC to consider.
How to submit your comments to us
Please send your responses to us by:
- Typing your comments into a Word document and attaching that to your email OR
- Typing your comments directly into your email.
Your comments can be as simple as a few bullet points or as long as two or three paragraphs.
Please email your comments to Rosemary Ainley at [email protected] no later than 5pm on Thursday 14 September, 2023.
We will collate your replies into a single submission and send it to the PBAC on your behalf.
If you wish to make your own submission directly to the PBAC you can do so by going to https://ohta-consultations.health.gov.au/ohta/pbac-nov-2023/




This Post Has 0 Comments